






Gain Access are lewis structures only for covalent bonds high-quality broadcast. No hidden costs on our binge-watching paradise. Become one with the story in a massive assortment of series made available in HD quality, optimal for discerning viewing fanatics. With hot new media, you’ll always be informed. Discover are lewis structures only for covalent bonds specially selected streaming in ultra-HD clarity for a truly engrossing experience. Register for our platform today to enjoy select high-quality media with 100% free, no strings attached. Get frequent new content and venture into a collection of bespoke user media produced for exclusive media lovers. Don't forget to get exclusive clips—instant download available! Get the premium experience of are lewis structures only for covalent bonds special maker videos with vibrant detail and unique suggestions.
Lewis structures for polyatomic ions follow the same rules as those for other covalent compounds. Lewis structures we also use lewis symbols to indicate the formation of covalent bonds, which are shown in lewis structures, drawings that describe the bonding in molecules and polyatomic ions For example, when two chlorine atoms form a chlorine molecule, they share one pair of electrons The lewis structure indicates that each cl atom has three pairs of electrons that are not used in bonding.
30 7.3 lewis structures and covalent compounds learning objectives by the end of this section, you will be able to Illustrate covalent bond formation with lewis electron dot diagrams Draw lewis structures depicting the bonding in simple molecules Lewis structures formation of covalent bonds can be represented using lewis symbols
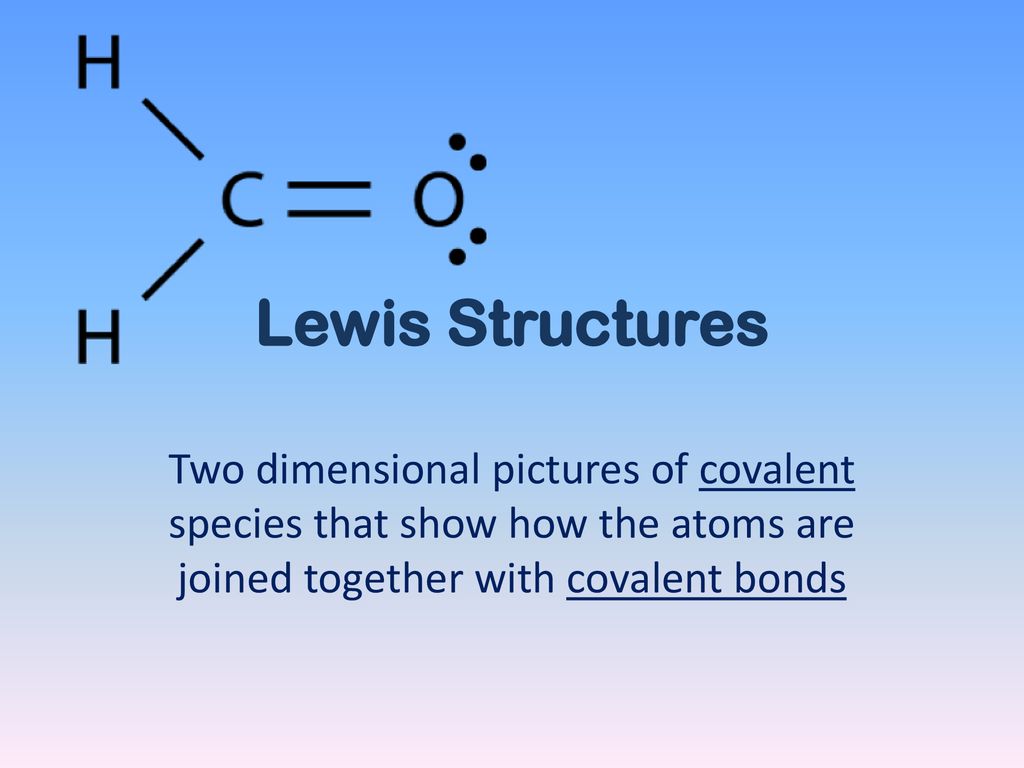
We usually show each electron pair shared between atoms as a line and show unshared electron pairs as dots Each pair of shared electrons constitutes one chemical bond. What is lewis structure lewis structure, also known as lewis dot structure or electron dot structure, is a simple and straightforward way of representing the outermost electron shell in a chemical species like an atom, ion, or molecule It shows how electrons are positioned around the atoms either as lone pairs or in a chemical bond, typically a covalent bond or a coordinate covalent bond.
The lewis structures illustrated so far have been selected for their simplicity A number of elaborations are given below There is sometimes an ambiguity in the location of double bonds This ambiguity is illustrated by the lewis structure for ozone (o3)
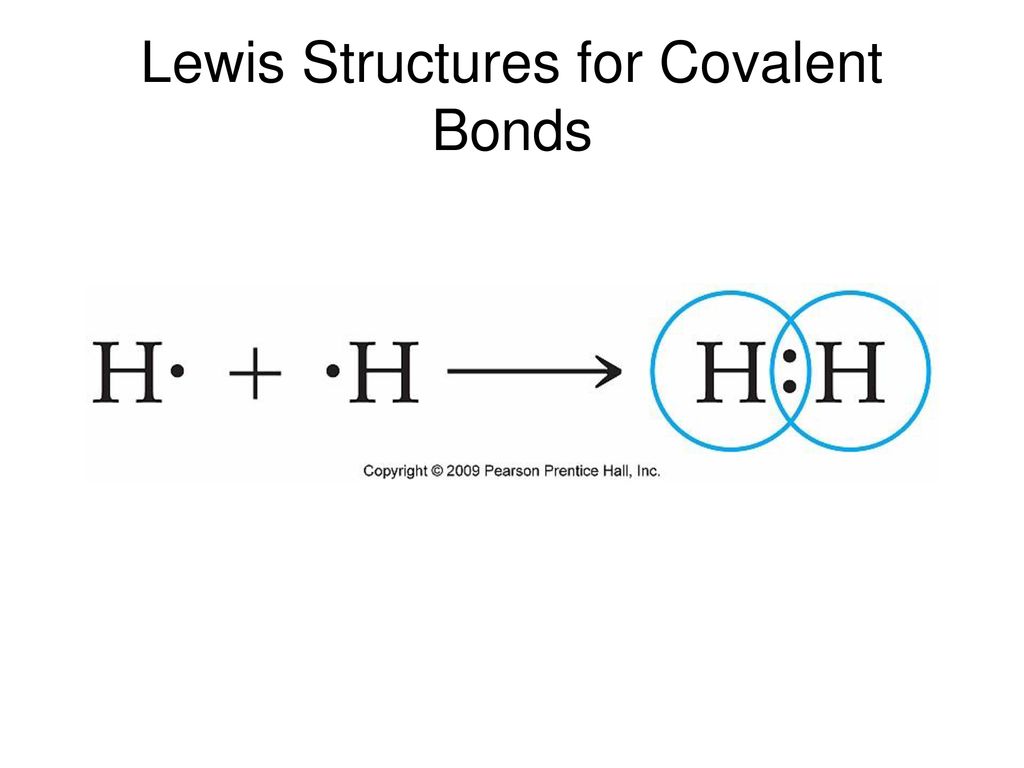
In such cases, the actual lewis. Lewis proposed that atoms combine in order to achieve a more stable electron configuration Maximum stability results when an atom is isoelectronic with a noble gas An electron pair that is shared between two atoms constitutes a covalent bond.

The Ultimate Conclusion for 2026 Content Seekers: Finalizing our review, there is no better platform today to download the verified are lewis structures only for covalent bonds collection with a 100% guarantee of fast downloads and high-quality visual fidelity. Don't let this chance pass you by, start your journey now and explore the world of are lewis structures only for covalent bonds using our high-speed digital portal optimized for 2026 devices. We are constantly updating our database, so make sure to check back daily for the latest premium media and exclusive artist submissions. Start your premium experience today!
OPEN